
In chemistry, another important thing to remember is that reduction occurs at cathode, and oxidation happens on anode, as what I mentioned above. Our conclusion is, elecctrons always move from anode to cathode in a circuit or electrolytic cell. Just like the cathode, the anode substrate is also coated with active material. 25kg of high purity graphite is needed for an average-sized battery, and up to 54kg for large batteries such as those used in the. which is porous in nature, usually graphite, as the anode, and metal oxide for the cathode 1524. Graphite is the most commonly used material for EV battery anodes. Energy density 40-110 Wh/kg at cell level. The substance is removed and can be blended with a small quantity of cobalt to create the cathode in Li-ion batteries. This is a rechargeable battery chemistry that has been superseded by lithium ion, but has seen a lot of use in Toyota hybrids. That means inside an electrolytic cell, eletrons move from anode to cathode. As such, the cathode plays a significant role in determining the voltage of the battery. The Nickel Metal Hydride battery has a nickel-hydroxide cathode, a metal hydride (a variety of metal alloys are used) anode, and aqueous potassium hydroxide electrolyte. Same thing happens inside the electrolytic cell.The anode is positively charged because cations (which are positively charged) moves to the anode from cathode, and the cathode is negatively charged because anions (which are negatively charged) moves to the cathode from anode.Cations gain electrons and are reduced at cathode, and anions lose electrons and are oxidize at anode. I will say yes, reason is that the electrons move from the anode to the circuit, then from the circuit back to the cathode. How does the electrons move inside and outside the circuit? Some people thinks that electrons move from anode to cathode. Lithium-ions and the electrons are transmitted through the electrolytes which then reunite at the cathode in a reduction reaction. 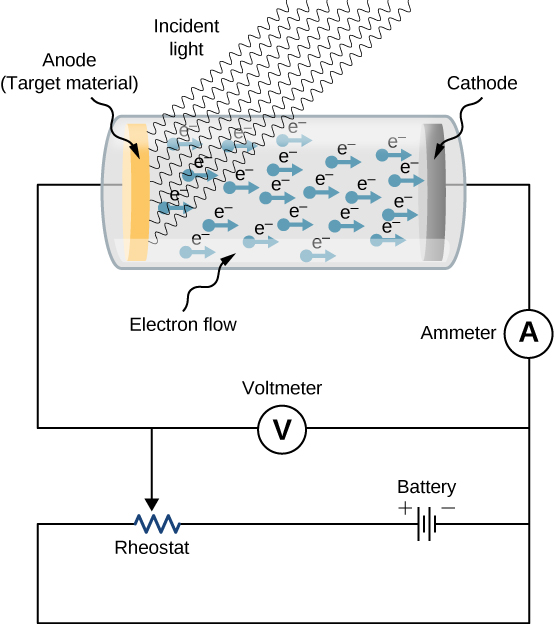
It is related to the single-wire system of the automobile, which means that the. Editors Thi Dieu Hien Nguyen, Shih-Yang Lin, Hsien-Ching Chung. 1, the grounding system: common in automotive technology, refers to the cars grounding system. First-Principles Calculations for Cathode, Electrolyte and Anode Battery Materials. 24 v battery cathode grounding often called iron, influential to the battery voltage is not large. That means the anode is positively charged, while the cathode is negatively charged. During the discharge phase of the battery, an oxidation reaction occurs at the anode which produces Lithium ions (positive), electrons (negative), and some by-products at the anode. Current from the battery anode flow, flow back to the battery cathode. For the regular alkaline battery we are using for example, we can see that the postitive end is anode, and the negative end is cathode. Some people are confused about cathode and anode when studying about chemistry or physics because of the appearance of battery.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
